Fluorescence-Guided Surgery for Brain Tumors: New Data
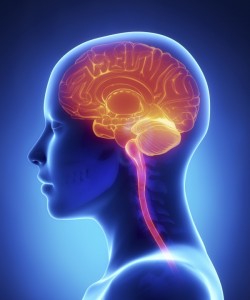
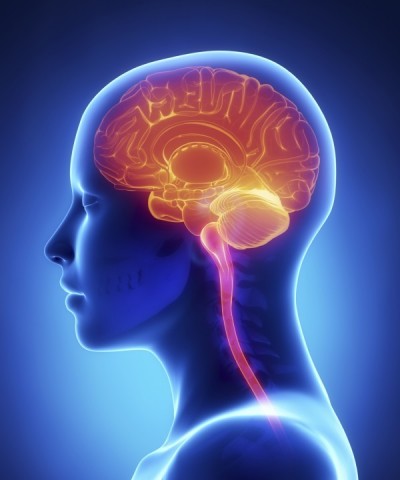
The term “brain cancer” refers to a group of malignant diseases that originates in the brain and spinal cord. The nerve cells in the brain are physically supported by the glial cells, which comprise the specialized connective tissue in the brain. Whereas neurons stop dividing almost completely after birth, glial cells continue dividing throughout life. This is why cancers of the glial cells—the so-called gliomas—can occur in adulthood.
Gliomas are the most common type of brain tumors, accounting for about six out of every ten primary malignant brain tumors, and they tend to be highly invasive. Brain tumors are also the second most common type of cancer diagnosed in children, though these tumors behave much less aggressively than those occurring in adulthood.
Mainstream treatment for brain cancer usually consists of surgery followed by radiation. The challenge for brain surgeons is to completely remove the tumor if this is feasible, while also striving to avoid major damage to the surrounding normal areas of the brain.
Unfortunately, full surgical removal of the tumor is technically not feasible due to the tumor’s ability to infiltrate or invade normal brain tissue. Cutting into the normal tissue can lead to considerable harm to the brain. In some situations, newer surgical techniques such as lasers may be tried. More advanced forms of laser therapy involve the use of photosensitizers that render the tumor-killing effects of the laser treatments far more selective, an approach known as photodynamic therapy, or PDT. A number of clinical trials have focused on the use of PDT in patients with GBM.
To help compensate for the limitations of conventional surgery, the approach known as fluorescence-guided surgery (FGS) has been developed. This procedure uses a photosensitizer to help reveal cancerous tissue during the surgery. By illuminating or “lighting up” any malignant tissue, surgeons can identify and remove smaller tumors that could otherwise be missed. They can easily see clusters of cancer cells as small as one-tenth of a millimeter, as opposed to the average minimal cluster size of 3 millimeters based on current methods of visual and tactile detection. As a relatively moderate-cost strategy,
FGS protocols have usually relied on the natural substance called 5-aminolevulinic acid. The substance is taken orally a few hours before surgery, leading to the selective concentration of the body’s very own photosensitizer—protoporphyrin IX or PpIX—within glioma cells. Under blue-violet light conditions, PpIX emits a reddish glow. (For more details about PpIX, please refer to the new book, The Medicine of Light.)
This deep red fluorescence reveals tumor tissue, while normal brain tissue appears to be colored blue. The clear contrast in colors enables the surgeon to then remove the malignant tissue, with a much smaller chance of removing or damaging healthy tissue. If any tumor tissue is too difficult to cut out (excise), it can then be treated with either PDT or radiotherapy. A number of clinical trials have sought to test the possibility that FGS along with repeated PDT treatments could be an effective strategy for advanced, recurrent GBM cases.
Researchers at Hospital de la Ribera in Valencia, Spain, recently tried using an exoscope (a microscope that can be easily viewed during the surgery) in order to perform FGS in 21 consecutive patients with high-grade glioma. A range of different brain tumors were included in the study: 14 patients had glioblastoma multiforme, two had grade-III oligodendrogliomas, one had anaplastic astrocytoma, three had metastases, and one had low-grade astrocytoma.
Total surgical removal of the tumors was achieved in 15 cases; subtotal resection was performed in 5 patients. None of the patients died as a result of the surgery. The researchers concluded that an exoscope can be used for FGS with 5-ALA. “With an important advantage of low cost, this allows the surgeon to perform collaborative surgeries and adds agility to the procedure,” they report in the April 2014 issue of the Austrian journal, Acta Neurochirurgica.
Finally, researchers from the Universidad Autónoma de Barcelona in Barcelona, Spain, evaluated the cost-effectiveness of 5-ALA-based FGS in patients with malignant glioma, under standard clinical practice conditions in Spain. They found that the incremental cost increase with 5-ALA versus conventional surgery using white light only amounted to € 4550 per additional complete remission achieved and € 9021 per quality-adjusted life year gained.
The investigators concluded that “malignant glioma surgery guided by 5-ALA fluorescence entails a moderate increase in hospital costs compared to current surgical practice and can be considered a cost-effective innovation,” as reported online ahead-of-print in the 24 January 2014 issue of Neurologia.
Support us by buying our book, The Medicine of Light, and ebooks from our Photoimmune Discoveries eBook Series.
Sources
Belloch JP1, Rovira V, Llácer JL, Riesgo PA, Cremades A. Fluorescence-guided surgery in high grade gliomas using an exoscope system. Acta Neurochir (Wien). 2014 Apr;156(4):653-60. Slof J1, Díez Valle R2, Galván J3. Cost-effectiveness of 5-aminolevulinic acid-induced fluorescence in malignant glioma surgery. Neurologia. 2014 Jan 24. Epub ahead of print] Smirnova ZS1, Ermakova KV, Kubasova IY, Borisova LM, Kiselyova MP, Oborotova NA, Meerovich GA, Luk’yanets EA. Experimental study of combined therapy for malignant glioma. Bull Exp Biol Med. 2014 Feb;156(4):480-2. © Copyright 2014, Photoimmune Discoveries, BV