Lighting Up Tooth Decay (Part 2)
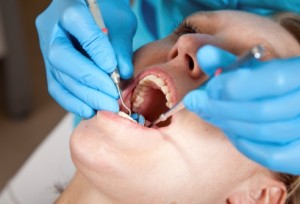
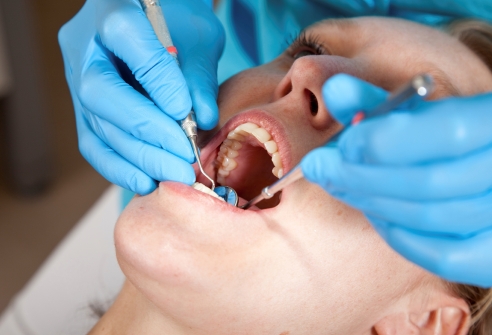
In Part 1, we introduced you to the problem of dental caries (cavities) and the role of bacteria in the mouth. Some of these bacteria stick to the tooth’s surface and convert the sugars or carbohydrates in our food into acids. These acids, in turn, cause the loss of calcium from tooth enamel—and the eventual formation of a cavity. Such cavities are undesirable because they provide a haven for the bacteria that may eventually damage the pulp deep within the gum, causing painful inflammation and eventually the loss of the tooth itself.
The bacterium called Streptococcus mutans (S. mutans) is the main causal agent for dental caries. To prevent this microbe from flourishing inside the mouth, dental professionals encourage people to limit their consumption of sugar and engage in good oral hygiene. The latter is achieved by brushing, flossing, teeth cleaning (by a dental hygienist), and in some cases the use of antimicrobial agents. All of these methods collectively aid in removal of the biofilm, more commonly known as plaque (see Part 1).
Alas, these methods also have some major limitations, such as damage to the lining the mouth caused by brushing or scraping methods. A more profound problem is the antibiotic resistance displayed by these microbes, particularly when they exist in the form of a biofilm. On a practical level, it is very difficult to maintain therapeutic levels of antibiotics inside the mouth.
Since dental caries are localized infections, alternative methods that by pass the problem of antibiotic resistance are needed. Alternative approaches are also needed because the mechanical treatments dentists use can sometimes expose the pulp, resulting in greater risk of periodontal disease.
Antimicrobial PDT: A Sensible Alternative
Recently, photodynamic therapy (PDT) has been introduced as a new modality for disinfecting the teeth. This approach is attractive in part because it preserves more of the dentin (a calcium-rich tissue in the tooth), thus enabling remineralization and preventing accidental pulp exposure. It also bypasses the problem of antibiotic resistance.
So-called antimicrobial PDT is a localized, non-thermal, and non-invasive method for disinfecting the tooth or at least greatly reducing bacterial colonies within the mouth. This entails a two-step protocol. First, the target cells such as the S. mutans microbes are exposed to a photosensitizer, the choice of which is extremely important.
In the second step, the microbes or plaque is exposed to harmless light of a specific wavelength. This results in the production of reactive oxygen molecules that can damage key components of the bacterial cells, such as the DNA and plasma membrane. The reaction can also alter metabolic activities in such a way as to cause cell death.
At this writing, antimicrobial PDT has several applications in dentistry such as the treatment of oral cancer as well as bacterial, viral and fungal infections. In addition, photodynamic diagnosis has been used to identify malignant lesions inside the mouth.
A number of studies have shown that antimicrobial PDT has a potent effect on a large number of oral gram-positive and -negative bacteria, using different photosensitizers and light sources. Nevertheless, the findings from these studies are not always clear or consistent. Due to these inconsistencies, scientists have begun to compare the effects of different PDT protocols by varying the photosensitizer and light sources.
Antibacterial Effects of Bremachlorin-PDT
In recent years, researchers from Tehran University’s Laser Research Center in Medical Sciences (Iran) have sought to assess the efficacy of antimicrobial PDT for reducing S. mutans colonies in the mouth. Most of these studies have been done in the laboratory, using standard cultures of S. mutans.
In 2011, the Tehran scientists showed that PDT had a pronounced effect against S. mutans when used with a helium-neon laser of a specific wavelength (633 nm). They concluded at the time that the photosensitizer Bremachlorin® was more effective with the laser than with either the laser or Bremachlorin® alone. This was a simple, elegant demonstration of the photodynamic principle: that it is the combination of light with a photosensitizer that spells the difference.
More recently, the Tehran researchers attempted to compare different PDT protocols in order to determine which one might be superior for eradicating the harmful microbes. In one protocol, they exposed S. mutans cultures to Bremachlorin in combination with laser light (662 nm). In a second protocol, they used Toluidine blue O (TBO) as the photosensitizer in combination with light-emitting diodes (630 nm). These protocols were tried separately as well as in combination. The control group for this study consisted of S. mutans cultures not exposed to any form of treatment.
The results were as follows. The combination of PDT with TBO and Bremachlorin® significantly reduced the viability of S. mutans. Intriguingly, however, there was no whereas no difference between the two PDT protocols. Moreover, there was no significant reduction of S. mutans colonies in the groups treated just with the photosensitizer or laser light alone. Thus, the S. mutans colonies were equally susceptible to either Bremachlorin-PDT or TBO-PDT, as reported in the April 2014 Journal of Applied Oral Science.
Finally, researchers from the University of Regensburg in Regensburg, Germany, are studying the possibility that positively charged phenalen-1-one derivatives can be used as a new generation of photosensitizer for PDT. This approach showed strong activity against not only S. mutans microbes, but also other oral key pathogens as well, including Enterococcus faecalis, Actinomyces naeslundii, and Aggregatibacter actinomycetemcomitans. These microbes were destroyed in less than one minute of light exposure after a single PDT treatment. Such a short exposure time could be especially advantageous for the antimicrobial treatment of dental caries, as reported in the 26 2014 Journal of Medicinal Chemistry.
Though the ideal combination of photosensitizer and light treatment remains to be discovered, we believe the photosensitizer known as Bremachlorin® holds great potential for antimicrobial PDT. It is just a matter of time before this agent becomes more widely used for treating infections and cancers alike. You can read more about recent research on Bremachlorin and in particular about exciting studies taking place in the Netherlands.
When you support Photoimmune Discoveries by buying our books and ebooks, you are supporting our educational efforts and indirectly helping to introduce such important medicines into the physician’s anti-cancer armamentarium.
Support us by buying our book, The Medicine of Light, and ebooks from our Photoimmune Discoveries eBook Series.
Sources
Späth A, Leibl C, Cieplik F, Lehner K, Regensburger J, Hiller KA, Bäumler W, Schmalz G, Maisch T. Improving photodynamic inactivation of bacteria in dentistry: highly effective and fast killing of oral key pathogens with novel tooth-colored type-II photosensitizers. J Med Chem. 2014 Jun 26;57(12):5157-68.
Fekrazad R, Bargrizan M, Sajadi S, Sajadi S. Evaluation of the effect of photoactivated disinfection with Radachlorin(®) against Streptococcus mutans (an in vitro study). Photodiagnosis Photodyn Ther. 2011 Sep;8(3):249-53.
© Copyright 2014, Photoimmune Discoveries, BV