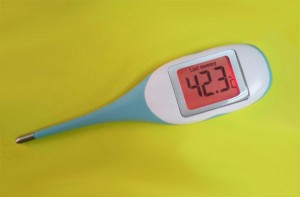
Heat, or increased body temperature, is a very old way of treating cancer, and some oncologists in Europe now consider it to be the “fourth” arm of mainstream treatment, or at least on a par with conventional immunotherapy. Hyperthermia treatments involves raising the temperature of the body (and thus the tumor tissue), usually to a range of 40-43°C. However, hyperthermia is not an appropriate stand-alone treatment option for most cancers. Instead, a number of clinical studies have found a clear benefit when hyperthermia is combined with other treatment modalities, in particular radiotherapy.
With radiation treatment, the heat is localized around the tumor area, rather than applied to the whole body. According to these studies, hyperthermia improves the effectiveness of radiotherapy for recurrent breast cancer and malignant melanoma, and results in increased survival for various groups of cancer patients (e.g., glioblastoma, cervical cancer, and head and neck lymph-node metastases). A more experimental approach is to use whole-body hyperthermia together with chemotherapy.
Another promising application of hyperthermia is to combine it with photodynamic treatment, or PDT. The basic principle is that PDT activates various immune cells that become more active against the cancer. Hyperthermia triggers the release of heat shock proteins (notably HSP-70), which further activate the body’s anticancer immune defenses. Experimental evidence suggests that the two treatments may work synergistically. To further boost the effectiveness of PDT, then, hyperthermia could be used as an adjunctive therapy, as suggested by the improved clinical outcomes seen in patients with rectal tumors who received both treatments simultaneously.
Hyperthermia also represents an effective option for restoring the chance of surgery for inoperable tumors and allows a new low-dosage treatment of relapsed cancers previously treated with high radiotherapy dosage without increasing toxicity. More recently, a new strategy has been developed that heats the tumor up from the inside using magnetic nanoparticles concentrated within the tumor site; a magnetic field is then applied to the tumor to achieve high core temperatures (42-45°C).